At the 2025 ASCO Annual Meeting, a phase 3 randomized trial revealed that the timing of immunotherapy in non-small cell lung cancer (NSCLC) significantly affects patient outcomes. This study, presented by Dr. Yongchang Zhang from Hunan Cancer Hospital, showed that infusions administered before 3 PM led to better progression-free survival (PFS) and overall survival (OS) compared to later infusions. Let’s dive into the findings and their implications for clinical practice.
Study Design: Randomized Trial on Timing of Immunotherapy in NSCLC
The study involved 210 patients with stage IV NSCLC, randomized into two groups of 105 each. One group received immunotherapy before 3 PM (Early ToD Group), while the other received it after 3 PM (Late ToD Group). Key eligibility criteria included driver gene-negative status, at least 18 years old, and no prior systemic therapy for metastatic disease. The trial aimed to assess whether the timing of immunotherapy influenced survival outcomes.
Key Findings: PFS and OS Results
The results highlighted significant differences between the two groups:
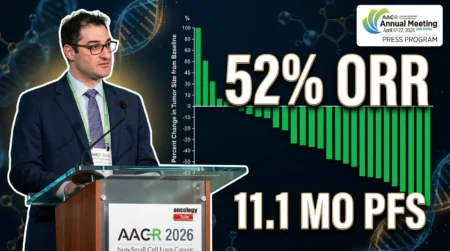
- Progression-Free Survival (PFS): The Early ToD Group achieved a median PFS of 11.3 months (95% CI, 9.3-13.3), compared to 5.7 months (95% CI, 3.1-4.5) in the Late ToD Group. The hazard ratio (HR) was 0.42, with a P-value less than 0.0001.
- Overall Survival (OS): The Early ToD Group had a median OS of 16.4 months (95% CI, 13.9-18.3), versus 10.9 months (95% CI, 8.0-13.8) in the Late ToD Group. The HR was 0.45, with a P-value less than 0.0001.
These findings suggest that administering immunotherapy earlier in the day may enhance its effectiveness in NSCLC patients.
Circadian Rhythms and T Cell Activity
The study also explored the biological basis for these results. Circadian rhythms were found to influence immune response, with circulating CD8+ T cells decreasing in the Late ToD Group (P<0.001). In contrast, the Early ToD Group showed increased activated (HLA-DR+) and TIM-3+PD-1+CD8+ T cells, indicating that morning infusions may optimize immune activity against cancer cells.
Safety Profile: Adverse Events Across Groups
Safety data showed no major differences between the groups. Grade 3-4 adverse events included:
- Leukopenia: 17.1% (Early ToD) vs. 21.0% (Late ToD)
- Neutropenia: 18.1% (Early ToD) vs. 22.9% (Late ToD)
- Pneumonitis: 2.9% in both groups
These findings indicate that adjusting the timing of immunotherapy does not significantly alter its safety profile.
Implications for Clinical Practice
With a median follow-up of 22.3 months, this study suggests that scheduling immunotherapy infusions earlier in the day could improve outcomes for NSCLC patients. This simple adjustment may also apply to other tumor types, offering a practical strategy for enhancing treatment efficacy without additional costs.
Explore the Slide Deck
For a deeper dive into the study, watch our video summary and explore the accompanying slide deck:
Learn More About NSCLC Research
Stay updated on the latest advancements in lung cancer treatment by visiting ASCO.org and OncologyTube.com.
For more insights into NSCLC, check out our related post:
https://oncologytube.com/category/non-small-cell-lung-cancer