Researchers continue to seek effective treatments for platinum-resistant ovarian cancer. At a recent medical conference, Tao Zhu from Zhejiang Cancer Hospital presented initial results from a first-in-human Phase 1 study of QHS-5132. These results are especially important for understanding QHS-5132 ovarian cancer therapies. This novel antibody-drug conjugate (ADC) targets Claudin 6. Moreover, QHS-5132 ovarian cancer research is rapidly evolving, offering new hope for patients.
A Highly Selective Target
Claudin 6 is a promising therapeutic target because it is rarely detectable in normal adult tissues. However, it is highly expressed in various malignancies, including ovarian and lung cancers. Furthermore, its expression often links to a poor prognosis.The investigational drug, QHS-5132, is a highly selective ADC. It consists of three parts: a humanized anti-Claudin 6 antibody, a stable linker, and a Topoisomerase I inhibitor payload. Additionally, it has a drug-to-antibody ratio of eight. In fact, QHS-5132 ovarian cancer treatments could potentially benefit many patients with limited options.
Phase 1 Study Design and Safety
The first-in-human study evaluated the safety and preliminary efficacy of QHS-5132 in patients with advanced solid tumors. Most participants had platinum-resistant ovarian cancer and had failed standard therapies. These patients were heavily pre-treated, with a median of three prior lines of therapy. The drug showed a favorable safety profile. The maximum tolerated dose was not reached. Most treatment-related adverse events were gastrointestinal or hematologic. These were generally Grade 1 or 2. “QHS-5132 was well tolerated with wider therapeutic windows, especially at a potential RP2D dose level of 4.8 and 5.6,” the speaker noted. Notably, QHS-5132 ovarian cancer safety data supports further trials.
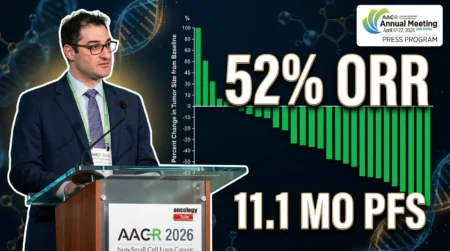
Promising Efficacy Regardless of Biomarker Status
The efficacy data showed significant potential. Across all dose levels, the overall response rate (ORR) was 50%. At the 4.8 mg/kg dose level, the ORR rose to 55.6%. Clinical activity occurred even in patients with low or undetectable Claudin 6 expression. This suggests the ADC might benefit a broad patient population. The speaker emphasized this advantage: “Our drug could be used for a much wider range of patients without mandatory screening.” While the sample size is currently small, the duration of response is encouraging. Some patients maintained their response for over six months. “Early data shows that anti-tumor activity over QHS-5132 did not depend strongly on Claudin 6 level,” the speaker explained. “This means the drug may benefit patients regardless of their biomarker status.” Enrollment is ongoing for cohorts focusing on non-small cell lung cancer, gastric cancer, and other tumors. In summary, QHS-5132 ovarian cancer therapies demonstrate robust efficacy.
Q&A for Busy Oncologists
What is the mechanism of action for QHS-5132? It is an antibody-drug conjugate (ADC) targeting Claudin 6. It delivers a Topoisomerase I inhibitor payload directly to cancer cells.
What were the efficacy results in platinum-resistant ovarian cancer? The study reported a 50% overall response rate across all doses. At the 4.8 mg/kg dose, the ORR was 55.6%.
Is Claudin 6 testing required for enrollment? Researchers tested primary tissue for Claudin 6, but patients enrolled regardless of their expression levels.
What were the main safety findings? The drug was well tolerated with no treatment-related deaths. Most side effects were Grade 1 or 2 gastrointestinal and hematological issues.