The management of KRAS G12D-mutant non-small cell lung cancer (NSCLC) has challenged clinicians for years. Doctors long relied on standard chemotherapy and immunotherapy, yet results often disappointed. The recent development and clinical investigation of Zoldenrasib KRAS G12D NSCLC is generating significant interest. New data from the RMC-9805-001 Phase 1 study now point to a potential breakthrough: zoldonrasib (RMC-9805), a first-in-class, oral, mutant-selective covalent inhibitor. For those following advances in lung cancer, Zoldenrasib KRAS G12D NSCLC represents an exciting development. Indeed, zoldonrasib KRAS G12D NSCLC research is rapidly evolving and attracting global attention.
In fact, zoldonrasib could soon change how oncologists treat KRAS G12D NSCLC.
Addressing a High Unmet Need
On the significant unmet need: “G12D is the most common oncogenic RAS mutation in human tumors and lacks an approved targeted treatment… This illustrates a large unmet need for targeted therapies in KRAS G12D lung cancer.” – Dr. Jonathan W. Riess, Associate Director of Clinical Research at the UC Davis Comprehensive Cancer Center
KRAS G12D drives many cancers. It affects roughly 61,000 new patients each year in the United States. In NSCLC, it appears in about 4% of cases—similar to the frequency of ALK fusions. Additionally, studies on zoldonrasib KRAS G12D NSCLC are particularly vital because of this high unmet need.
Oncologists must identify these patients quickly. They often show a distinct clinical profile:
- Smoking history: Many are never- or light-smokers (45%–56% in study cohorts).
- Biomarkers: These tumors typically have lower tumor mutational burden (TMB) and lower PD-L1 expression than other KRAS mutations.
- Current limitations: After progression on platinum-based chemotherapy plus immune checkpoint inhibitors (ICIs), second-line docetaxel delivers only 3–4.5 months median progression-free survival.
Does Zoldenrasib Show Strong Clinical Efficacy?
Zoldonrasib targets the active GTP-bound “ON” state of the KRAS G12D protein. It functions as a molecular glue. This action forms a tri-complex that blocks RAF interaction and halts downstream oncogenic signaling. Notably, the efficacy seen in Zoldenrasib KRAS G12D NSCLC trials is driving more research.
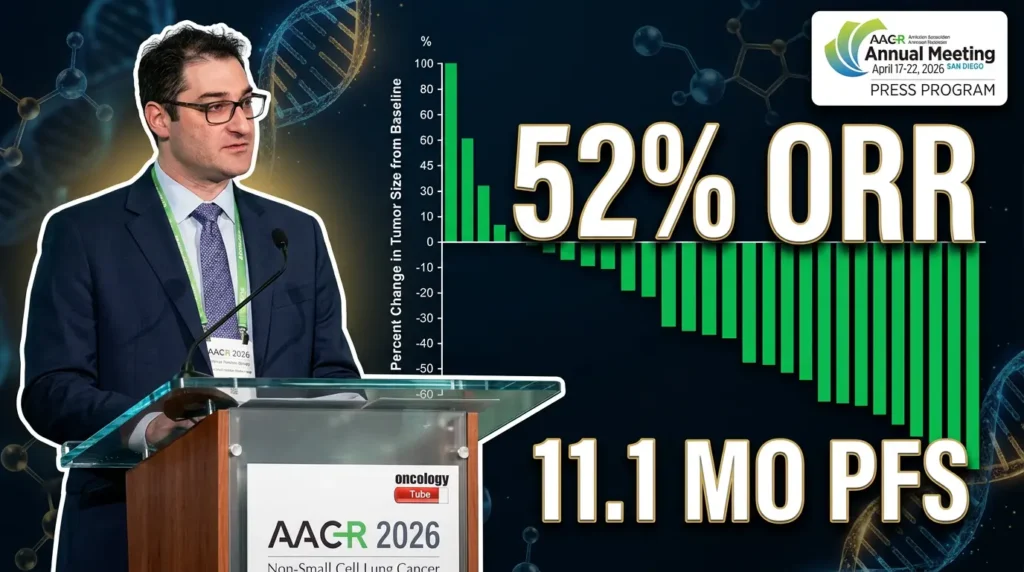
Researchers evaluated zoldonrasib at the recommended Phase 2 dose (RP2D) of 1,200 mg once daily. The efficacy population included 27 previously treated patients (all received prior platinum chemotherapy and ICIs). Key results included:
- Confirmed overall response rate (ORR): 52%
- Disease control rate (DCR): 93%
- Median progression-free survival (PFS): 11.1 months
- 12-month overall survival (OS): 73%
- Molecular response: 73% of evaluable patients achieved complete clearance of KRAS G12D circulating tumor DNA (ctDNA)
On the drug’s mechanism of action: “Zoldenrasib is an oral, mutant-selective, covalent inhibitor targeting the active, GTP-bound state of KRAS G12D… It acts as a ‘molecular glue’ binding inside the cell to cyclophilin A to form a tri-complex that sterically occludes RAF interaction.” – Dr. Jonathan W. Riess
Is the Safety Profile Suitable for Combinations?
Zoldonrasib stands out for its favorable safety profile. Patients maintained a median dose intensity of 97%. Most treatment-related adverse events (TRAEs) stayed Grade 1 and remained manageable. Therefore, Zoldenrasib KRAS G12D NSCLC patients may benefit from early and ongoing combination studies. Overall, as more data accumulate, the future of zoldonrasib KRAS G12D NSCLC treatments appears bright.
Common side effects included:
- Gastrointestinal: nausea (43%), vomiting (33%), diarrhea (30%)
- Dermatological: Grade 1 rash (18%)
- Hepatic: Grade 1 AST increase (10%)
Importantly, clinicians reported no Grade 4 or 5 TRAEs. Only 5% of patients discontinued treatment due to adverse events. This clean profile—even in patients with prior immunotherapy—supports testing zoldonrasib in earlier treatment lines. It also encourages combination studies with chemo-immunotherapy or other RAS inhibitors, such as daraxonrasib (note: the agent previously referenced as dudaraxinrasib is daraxonrasib / RMC-6236).
On the clinical results: “Zoldenrasib has demonstrated encouraging clinical activity at the recommended Phase 2 dose… with a confirmed overall response rate of 52% and a median progression-free survival of 11.1 months, comparing extremely favorably to the second-line standard of care.” – Dr. Jonathan W. Riess
Conclusion
Early data look promising. Zoldonrasib delivered a 52% confirmed response rate and nearly tripled median PFS compared with historical second-line standards. It may become a new cornerstone of targeted therapy for KRAS G12D NSCLC. As research continues, Zoldenrasib KRAS G12D NSCLC could transform patient outcomes in this challenging disease subset. In summary, zoldonrasib KRAS G12D NSCLC represents one of the most promising advances for patients with this mutation.
Q&A for Busy Oncologists and Researchers
Q: How does zoldonrasib’s mechanism differ from first-generation KRAS inhibitors? A: First-generation inhibitors bind the inactive GDP-bound “OFF” state. In contrast, zoldonrasib acts as a molecular glue on the active GTP-bound “ON” state. It forms a tri-complex with cyclophilin A that sterically blocks RAF interaction and shuts down oncogenic signaling.
Q: What is the clinical significance of the patient demographic in this study? A: About 50% of participants were never-smokers. This matches the typical KRAS G12D NSCLC profile: light or never-smoking history, lower TMB, and lower PD-L1 expression.
Q: Does the safety profile support combination therapy? A: Yes. Low rates of hepatic issues (only 10% Grade 1 AST increase) and no Grade 4/5 events suggest zoldonrasib can combine safely with immunotherapy or platinum-based chemotherapy in earlier lines. Studies are also exploring pairings with multi-selective RAS inhibitors like daraxonrasib. In the context of innovation, Zoldenrasib KRAS G12D NSCLC is being watched closely by the oncology community.
Q: Is there evidence of CNS activity? A: Data remain limited. The study allowed patients with treated, stable, asymptomatic brain metastases (about 25% of the safety population) but excluded active brain metastases. More research is needed to assess CNS efficacy.