Introduction
Zongertinib (BI 1810631) has shown promising results in treating HER2-mutant non-small cell lung cancer (NSCLC), with a 71% response rate as presented at AACR 2025. This oral, HER2-selective tyrosine kinase inhibitor (TKI) offers a new option for patients with this challenging condition. In this post, we explore the Beamion LUNG-1 trial results, zongertinib’s clinical benefits, and its potential impact on NSCLC treatment protocols.
Zongertinib’s Efficacy in HER2-Mutant NSCLC
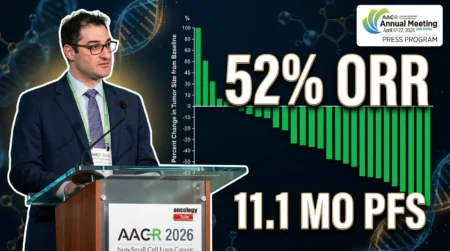
The Beamion LUNG-1 trial revealed zongertinib’s impressive efficacy in previously treated HER2-mutant NSCLC patients. It achieved a 71% objective response rate (ORR). This means 71% of patients experienced tumor shrinkage or disappearance. Additionally, the median progression-free survival (PFS) reached 12.4 months. This indicates a significant delay in disease progression. For patients with brain metastases—a common issue in this subtype—zongertinib demonstrated a 41% intracranial ORR. It offers hope for addressing this critical complication.
Zongertinib also showed a median duration of response (mDOR) of 14.1 months, highlighting the durability of its effects. These results were presented at the 2025 American Association for Cancer Research (AACR) Annual Meeting. They were also published in the New England Journal of Medicine, underscoring their credibility and relevance in the oncology community.
FDA Priority Review and Future Potential
In February 2025, the FDA granted priority review to zongertinib for the treatment of unresectable or metastatic HER2-mutant NSCLC in patients who have received prior systemic therapy. A decision is expected in Q3 2025, potentially accelerating its availability for clinical use. The ongoing BEAMION LUNG-2 trial is evaluating zongertinib as a frontline therapy. If successful, this could expand its role in NSCLC treatment protocols.
Safety Profile: A Comparison with Trastuzumab Deruxtecan
Zongertinib’s safety profile is a key advantage. Compared to trastuzumab deruxtecan, a commonly used HER2-targeted therapy, zongertinib had fewer severe side effects. Grade 3 or higher adverse events (AEs) occurred in only 17% of patients on zongertinib, versus 40% for trastuzumab deruxtecan. Notably, zongertinib reported no cases of interstitial lung disease (ILD), a serious side effect associated with some NSCLC treatments. Its EGFR-sparing mechanism also reduces common side effects like rash and diarrhea, improving patient tolerability.
Mechanism of Action
Zongertinib is an oral, irreversible HER2-selective TKI designed to target HER2 mutations in NSCLC. By sparing EGFR signaling, it minimizes toxicity while effectively inhibiting HER2-driven tumor growth. This targeted approach makes it a promising option for patients with HER2-mutant NSCLC, particularly those who have progressed on prior therapies.
Expert Perspectives from AACR 2025
At AACR 2025, Dr. John Heymach from MD Anderson Cancer Center presented the Beamion LUNG-1 trial results, sparking significant interest. Dr. Stephen Liu noted the publication on X, emphasizing zongertinib’s efficacy and tolerability.
Why Zongertinib Matters for NSCLC Patients
Zongertinib may provide a new treatment option for HER2-mutant NSCLC patients, especially those with brain metastases. Its ability to achieve a 41% intracranial ORR addresses a critical need, as brain metastases are a common challenge in this subtype. With fewer toxicities and no reported ILD, zongertinib offers a safer alternative to existing therapies. It potentially improves patients’ quality of life.
Related Links
https://www.nejm.org/doi/full/10.1056/NEJMoa2503704