Published: June 4, 2025
Category: Lung Cancer Research
Introduction to Neoadjuvant Nivolumab Plus Chemotherapy in CheckMate 816
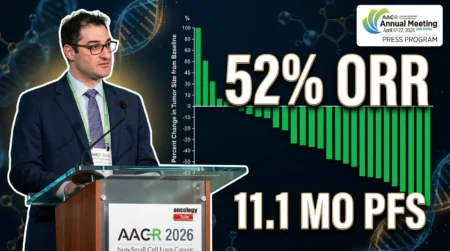
The CheckMate 816 trial offers new findings on treating resectable lung cancer with neoadjuvant nivolumab plus chemotherapy. For example, it was presented at ASCO 2025 and published in the New England Journal of Medicine. Therefore, this study shows how neoadjuvant nivolumab plus chemotherapy in CheckMate 816 can help patients with non-small-cell lung cancer (NSCLC). Dr. Patrick Forde and his global team led the research. Watch the video below for a quick summary of the results. It’s tailored for oncologists and researchers.
Video: CheckMate 816 Trial Update
Video Description: This 60-second video summarizes the CheckMate 816 trial. It highlights five-year survival data, response rates, and safety for neoadjuvant nivolumab plus chemotherapy in NSCLC. The video was presented at ASCO 2025.
Key Findings from the CheckMate 816 Trial
The CheckMate 816 trial tested neoadjuvant nivolumab plus chemotherapy in NSCLC patients. Here are the main results:
- Five-Year Survival Rates: Neoadjuvant nivolumab plus chemotherapy led to a 65% survival rate at five years. In contrast, chemotherapy alone achieved 55% (hazard ratio: 0.73).
- Pathological Complete Response (pCR): 24% of patients in the nivolumab group had a complete response. As a result, they showed an 88% event-free survival rate at five years.
- PD-L1 Subgroup Analysis: Patients with PD-L1 levels of 1% or higher had a 71% survival rate with neoadjuvant nivolumab plus chemotherapy.
- Safety Profile: Adding nivolumab to chemotherapy did not increase toxicity. Additionally, it did not affect surgery feasibility, making it a safe option.
These results, published in the NEJM, suggest better outcomes for lung cancer patients with neoadjuvant nivolumab plus chemotherapy.
Why This Matters for Oncologists
This trial provides useful data for oncologists and researchers. For instance, the improved survival rates show the potential of neoadjuvant nivolumab plus chemotherapy in CheckMate 816 before surgery. Moreover, the high complete response rates are encouraging. The safety data also confirms that this treatment can be used in practice. Thus, it’s a promising approach for resectable lung cancer.
Explore the Full Study and Join the Discussion
You can read the full study in the New England Journal of Medicine. Here’s the link: nejm.org/doi/full/10.1056/NEJMoa2502931. Also, join the discussion at ASCO 2025 to learn more about lung cancer treatment with neoadjuvant nivolumab plus chemotherapy. Share your thoughts in the comments or on social media with #CheckMate816 and #ASCO25.
Conclusion
The CheckMate 816 trial is an important step in lung cancer research. It shows better survival and safety with neoadjuvant nivolumab plus chemotherapy. Therefore, this study sets the stage for future advancements in NSCLC treatment. Stay updated with OncologyTube.com for more oncology research news.
Related Posts: